Good news: Disposable Introducer Sheath obtained Class II Medical Device Registration certificate
On June 20, Lepu ScienTech Medical Technology (Shanghai) Co., Ltd. ("ScienTech Medical") has achieved another milestone with its self-developed product, the "Disposable Introducer Sheath," obtained the Class II Medical Device Registration Certificate (Approval No. 20232030162) following the official approval of the "G-cruiser® Interventional Delivery System for Biodegradable Occluder" on June 19. This product serves as a percutaneous access channel for intravascular devices, and offers clinicians a wider range of treatment options to meet diverse clinical needs.
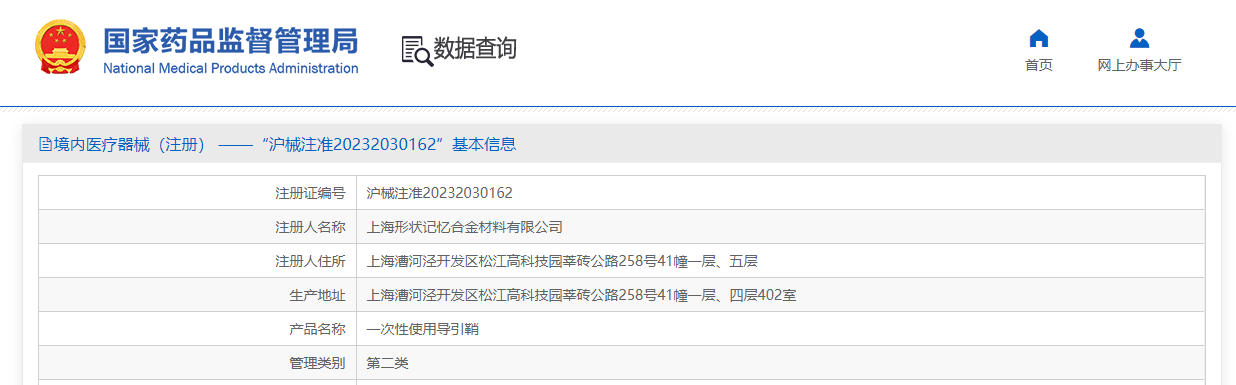
(Source: Official Website of the National Medical Products Administration)
Precise Placement and Stable Attachment
The interventional procedures has became the primary approach for treating cardiovascular, cerebrovascular, and peripheral vascular problems. It depend on vascular puncture to establish appropriate access points. The disposable introducer sheath developed by ScienTech Medical serves as an auxiliary instrument for peripheral vascular puncture in cardiovascular and vascular interventional surgery by use of the Seldinger vascular puncture technique. It has a multi-layered sealing pad design that ensures smooth device delivery while effectively controlling bleeding. The hydrophilic coating on the surface of the sheath and dilator facilitates smooth passage without causing damage to the entry vessel and reduces the risk of bleeding. Stable punctures are guaranteed by the rotating lock that firmly secures the sheath and dilator at the proximal end. It is possible to make safer delivery by the improved proximal design, which enables a snug, seamless transition with little resistance.
Additionally, this introducer sheath is available in a variety of specifications to meet a wide range of clinical requirements with lengths ranging from 33 cm and 45 cm to 65 cm, and diameters from 12 to 24F. The 12F size is designed to fulfill the specific clinical needs of narrower peripheral vessels, particularly in cases of lower limb vascular diseases. Additionally, the 45cm and 65cm long sheaths are exceptionally well-suited for patients with tortuous or angulated vessels located above the diaphragm.
Explore Diligently, Grow Stronger
The approvals for two innovative products in a single week not only highlights the competence in research and innovation capabilities of ScienTech Medical but also elevates the level of medical care offered to clinical doctors and patients.
In the future, ScienTech Medical will persist in harnessing its technological expertise to pursue ongoing research and innovation. ScienTech Medical will explore diverse medical application scenarios and further into the area of structural heart disease. With a commitment to support the clinical community and larger patient population, the ScienTech Medical's objective is to introduce innovative products that are not only of superior quality but also safer and more effective.

-

ScienTech Medical was awarded the second prize of the National Technological Innovation Award
2024-07-03 -

Exhilarating news: ScienTech Medical’s MemoSorb® Biodegradable Patent Foramen Ovale (PFO) Occluder obtained NMPA approval
2023-09-17 -

ScienTech Medical reveals impressive one-year follow-up results of ScienCrown® Valve System for TAVR clinical study at GW-ICC2023
2023-09-13
Copyright © 2021, Scientech Medical. All rights reserved. 沪公网安备31011702008238号沪ICP备2021017431号
沪公网安备31011702008238号沪ICP备2021017431号







